상온에서 가능한 탄소-수소 결합촉매반응From C-H to C-C Bonds at Room Temperature
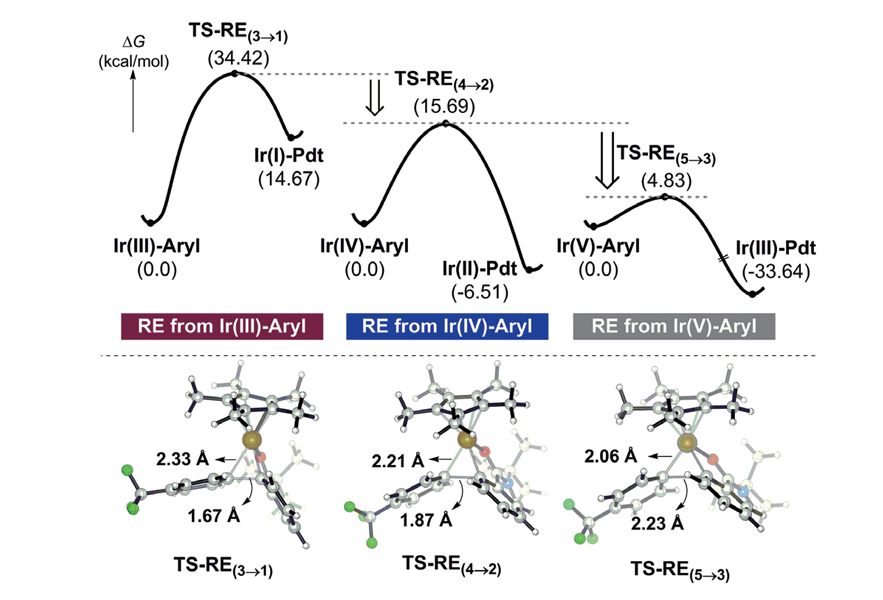 탄화수소는 자연상태에 많이 존재하지만 일반적 조건에서는 반응성이 낮아 합성의 원료로 사용되기 어렵다. 본 연구는 합성실험과 계산화학연구를 바탕으로 지금까지 불가능 하다고 여겨졌던 값싼 탄화수소의 아릴화 반응을 상온에서 이리듐을 이용해서 성공했다. 이 새로운 방법을 통해 의약품 및 재료 물질과 같은 중요한 화합물을 쉽고 빠르게 합성할 수 있을 것으로 기대된다.
탄화수소는 자연상태에 많이 존재하지만 일반적 조건에서는 반응성이 낮아 합성의 원료로 사용되기 어렵다. 본 연구는 합성실험과 계산화학연구를 바탕으로 지금까지 불가능 하다고 여겨졌던 값싼 탄화수소의 아릴화 반응을 상온에서 이리듐을 이용해서 성공했다. 이 새로운 방법을 통해 의약품 및 재료 물질과 같은 중요한 화합물을 쉽고 빠르게 합성할 수 있을 것으로 기대된다.Direct arylation of C–H bonds is in principle a powerful method of preparing value-added molecules that contain carbon-aryl fragments. Unfortunately, currently available synthetic methods lack sufficient effectiveness to be practical alternatives to conventional cross-coupling reactions. This study hypothesized that the main problem lies in the late portion of the catalytic cycle in which reductive elimination produces the desired C–C bond. Based on this mechanistic hypothesis, a new strategy was implemented where the Ir(III) center of the key intermediate is oxidized to Ir(IV). Density functional theory calculations indicate that the reductive elimination barrier is reduced by almost 19 kcal/mol when the metal is oxidized. Various experiments confirmed this prediction, offering a new methodology capable of efficiently and directly arylating C–H bonds at room temperature with a broad substrate scope and good yields. This study highlights how the oxidation states of intermediates can be deliberately targeted to rationally catalyze an otherwise impossible reaction.
- Description
- 한국과학기술원 : 화학과
- Publisher
- 한국과학기술원
- Issue Date
- 2018
- Language
- kor
- Description
KAIST 2018 대표 연구성과 10선
- Link
- https://archives.kaist.ac.kr/research.jsp?year=2018&view=view02
https://archives.kaist.ac.kr/eng/research.jsp?year=2018&view=view02
- Appears in Collection
- 2018 KAIST 대표 연구성과 10선
Items in DSpace are protected by copyright, with all rights reserved, unless otherwise indicated.



